 Oxygen is a nonpolar diatomic molecule with a 180-degree bond angle.īoth oxygen atoms in the molecule have the same electronegativity value, and both atoms share identical ratios of bound shared electrons, making the O2 molecule nonpolar in nature. 
In the O2 Lewis structure, there is a double bond between two oxygen atoms. They can be identified by using a Lewis structure. Lone pair of electrons are found in the outermost electron shell of atoms. The lone pair of electrons are unshared valence electrons. Oxygen also possesses a lone pair of electrons. Similarly, to complete its octet, oxygen shares three valence electrons with carbon. By sharing three valence electrons, carbon forms three tipple bonds with oxygen. The overall carbon to oxygen atom ratio in a CO dot structure is 1:1. Is Carbon monoxide acidic or basic?Ĭarbon monoxide is a neutral gas since it does not show basic and acidic properties when it reacts with water 2. If you have any questions related to this blog, please feel free to comment. The following are some of the frequently asked questions. Related Links N 2O Lewis Structure HCN Lewis Structure SiO 2 Lewis Structure SO 2 Lewis structure BF3 Lewis structure| Molecular geometry, Hybridization Frequently Asked Questions (FAQs) Each carbon and oxygen atom contains one lone pair of electrons.In the CO molecule, C-O length is 112.8 pm.Carbon monoxide molecules consist of a carbon atom covalently double bonded to the oxygen atom.Carbon monoxide is the most common type of fatal poisoning in the world.CO is a highly toxic gas and can cause acute illness and, in worst-case scenarios, death.It is produced by the incomplete burning of fossil fuels.It is highly poisonous, odorless, colorless, and tasteless.Carbon and oxygen from one sigma bond and two pi bonds. CO Molecular GeometryĬarbon monoxide has a linear molecular geometry, a triple bond between C and O, and each atom contains one lone pair of electrons. To complete the octets of both atoms, place two valence electrons on both atoms. This will leave four valence electrons only. In contrast, oxygen has six valence electrons and it needs two more electrons to complete the octet.Ĭonstruct three bonds between oxygen and carbon atoms. The last step is to complete the octet of the molecule.Ĭarbon has four valence electrons and it needs four more to complete its octet. VEs in carbon monoxide = VEs in 1 carbon atom + VEs in 1 oxygen atom To draw the Lewis structure, we need to figure out the number of valence electrons in individual atoms as shown in the table. Step-1: Count the valence electrons of atoms Oxygen has one lone pair of electrons as well. Similarly, oxygen shares three valence electrons with carbon to complete its octet. Carbon forms three triple bonds with oxygen by sharing three valence electrons. So this is correct.In a CO Lewis structure, the overall ratio of carbon to the oxygen atom is 1:1. Here, carbon is less unhappy both atoms have octets, and the electrons are more evenly distributed, minimizing the formal charges AND minimizing the energy overall in this major resonance structure. 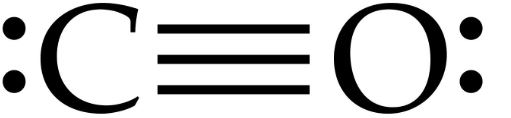
Finally, we land upon the only other rational possibility: The tension of the unhappy oxygen really wanting the electrons destabilizes this particular resonance structure.īut carbon does not have an octet, so this is not realistic. 
Carbon is less electronegative than oxygen, so it won't be happy with having more electrons than oxygen. With #10# electrons, one might predict this structure:īut that is not likely correct. #"Charge = valence electrons - owned electrons"# Assign formal charges, and fix the resonance structure by moving electrons and bond lines around until the formal charges are minimized.(There are two #pi# bonds and one #sigma# bond in a triple bond, one #sigma# and one #pi# bond in a double bond, and one #sigma# bond in a single bond.) Where there are electron pairs, construct one bond line for each electron pair.Place all electrons in predicted spots.Draw out a predicted atom connectivity.Count the number of valence electrons per atom.Students are typically taught an electron-counting method, which goes as follows: This often looks wrong to a student who is used to seeing double bonds on oxygen. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
